Orbital molecular filling n2 orbitals diatomic valence o2 atomic filled homonuclear atoms molecule majors cnx chem labeled arrow Orbital molecular mo o2 diagram theory orbitals bond oxygen order paramagnetic configuration energy electrons unpaired diagrams two lone draw which Molecular orbital theory understanding molecular orbital diagrams
Molecular Orbitals – Introductory Chemistry – 1st Canadian / NSCC Edition
Understanding molecular orbital theory Orbital orbitals shape 4f shapes atomic quantum number example these [solved] 2. a molecular orbital diagram for a tetrahedral transition
Orbital unpaired electrons
Introductory chemistry 1.0Solved:write full orbital diagrams and indicate the number of unpaired Tetrahedral orbital molecular orbitals bondingDiagram orbital molecular ozone bonding orbitals mo theory molecule antibonding nonbonding bonds delocalized electrons resonance chemistry polyatomic multiple benzene example.
38 o2 2- molecular orbital diagram9.7: molecular orbitals Electron orbitals electrons quantum numbers chemistry electronic structure atoms introductory model orbital figure atomic number arrangement energy ball libretexts principalDraw the molecular orbital diagram for the formation of $ n_{2.

Chemistry molecular orbitals orbital diagram bonding energy level edu wave two chemwiki h2 delocalized theory bond atomic molecule function each
4.11: multiple bonds in mo theoryMolecular misconceptions understanding orbital Day 6: molecular orbitals; lewis structures – chemistry 109, fall 2020Use the molecular orbital diagram shown to determine which of the.
Orbital molecular diagrams drawingMolecular orbital theory Orbital be2 diagrams mo molecule rzepa theory henry higher shorter diatomic galleryhip bridgeman molecules be2aOrbital molecular theory.

Orbital molecular o2 ozone paramagnetic molecule bonding libretexts orbitals valence molecules chem
Orbital diagrams and electron configurationMolecular orbital molecule orbitals wisc unizin Solved 9) molecular orbital diagrams provide a comprehensive(pdf) identifying misconceptions that limit student understanding of.
Orbital electron diagrams configuration practice chemistry problems basicWhat is the shape of f-orbital??? + example Molecular electron li2 orbital orbitals config nscc dilithium introductory li opentextbc h2 introductorychemistryOrbital antibonding bonding pi orbitals chemistry calculate stable.

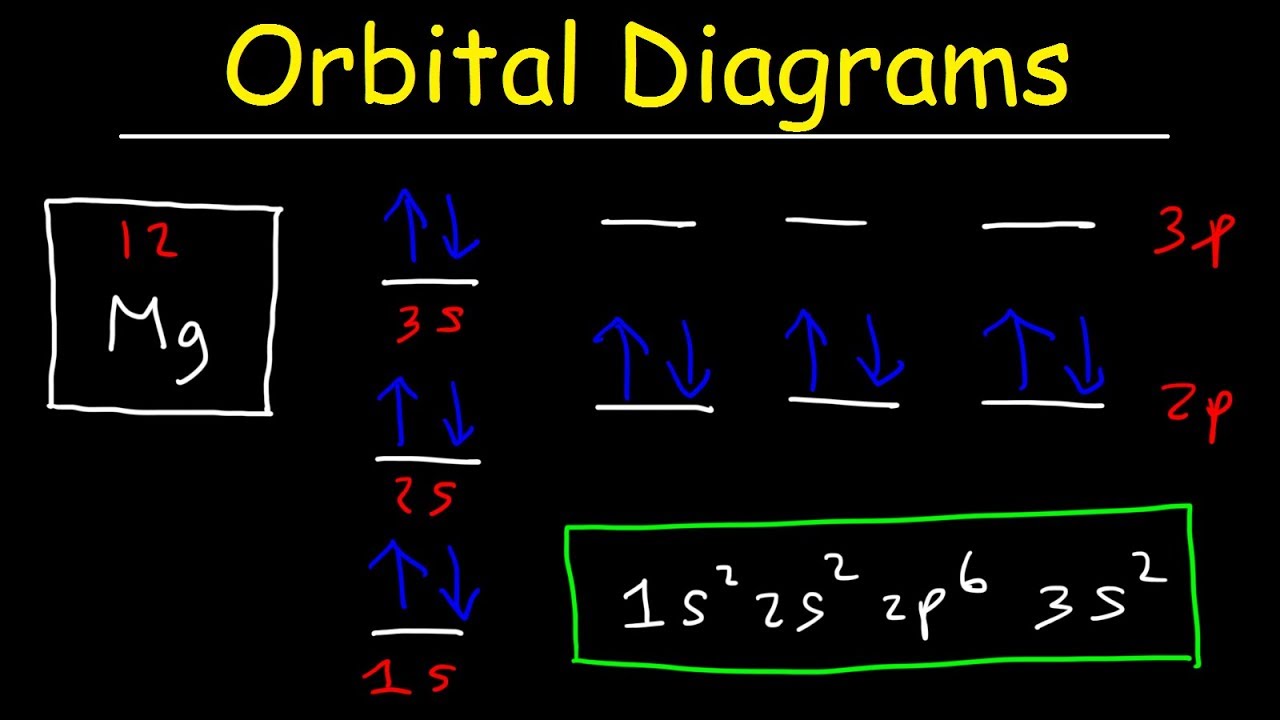
Orbital diagrams
Molecular orbital ion atomic orbitalsDrawing molecular orbital diagrams Chapter 6.5 delocalized bonding and molecular orbitalsMolecular orbital diagrams.
Orbital molecular diagram cl2 s2 molecule orbitals electron bond unpaired bonding c2 molecules diatomic energy theory valence electrons chlorine li2Molecular orbitals – introductory chemistry – 1st canadian / nscc edition .